Last Updated:
February 24, 2026 This Has Been Corrected / Updated. View Update HistoryAuthors:
Shawn D. St. Peter*, Krow Ampofo*, Thomas Brogan, Michael D. Cabana, Claudia Espinosa, Todd A. Florin, Jeffrey S. Gerber, Michelle Gill, Debra L. Palazzi, Mark Sawyer, Angela M. Statile, Derek J. Williams, Sheena Patel*, Mark I. Neuman*, Samir S. Shah*Related Content:
IDSA/PIDS 2011 Guidelines for the Management of Community-Acquired Pneumonia (CAP) in Infants and Children Older Than 3 Months of AgeKeywords:
pneumonia, parapneumonic effusion, empyema, guidelineBackground
The last iteration of this guideline was published in 2011 and a need to incorporate contemporary evidence prompted an update. The scope of this guideline includes the diagnosis and management of pneumonia with parapneumonic effusion in children.
Complicated pneumonia, including parapneumonic effusion, empyema, lung abscess, and necrotizing pneumonia, occurs in a small portion of children with pneumonia, and is associated with higher rates of morbidity and mortality. Estimates of the rates and prevalence of complicated pneumonia vary, but one study observed that 7.9% of children <2 years and 16.9% of children 2-4 years of age with pneumococcal pneumonia had empyema [Grijalva].
For the purposes of this guideline, complicated pneumonia is defined as pneumonia associated with parapneumonic effusion and/or empyema. A parapneumonic effusion is fluid accumulation in the pleural space related to pneumonia and may be free-flowing and sterile or complicated (infected or markedly inflammatory). Effusions are classified as small, moderate, or large based on radiographic extent. Empyema refers to purulent fluid in the pleural space associated with pneumonia.
This guideline addresses the diagnosis and management of pediatric pneumonia with parapneumonic effusion. It excludes populations in low-resource settings, where diagnostic and management approaches differ. The guideline is intended for healthcare professionals involved in the care of children with pneumonia and complicated pneumonia, including clinicians in infectious diseases, emergency medicine, pulmonology, hospital medicine, surgery, and critical care.
Methods
The panel included pediatric clinicians with expertise in infectious diseases, emergency medicine, hospital medicine, general pediatrics, and general surgery. Panelists were diverse in gender, geographic distribution, and years of clinical experience. Selected reviewers included clinicians with expertise in pediatric infectious diseases. The following organizations reviewed and endorsed the guideline: The Pediatric Infectious Diseases Society (PIDS).
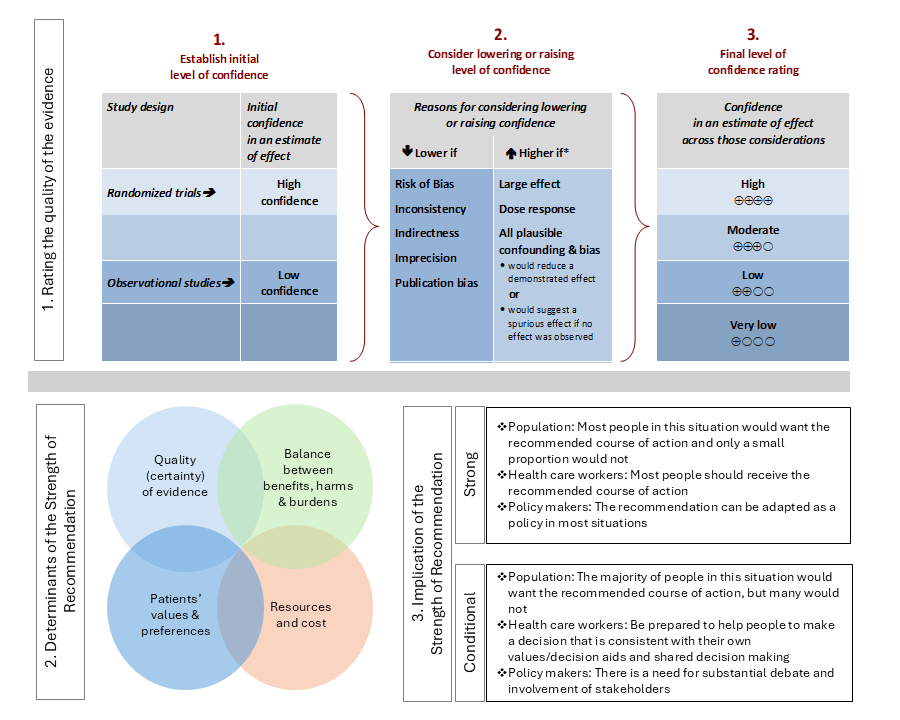
For each question, a systematic review was performed to identify relevant studies and the GRADE (Grading of Recommendations Assessment, Development and Evaluation) approach was followed for assessing the certainty of evidence and strength of recommendation (Figure 1).
Details of the systematic review and guideline development process for each recommendation are available in the supplemental materials for each included manuscript.
Figure 1. Approach and implications to rating the quality of evidence and strength of recommendations using GRADE methodology (unrestricted use of figure granted by the U.S. GRADE Network)
Reference
- Grijalva CG, Nuorti JP, Zhu Y, Griffin MR. Increasing incidence of empyema complicating childhood community-acquired pneumonia in the United States. Clin Infect Dis. 2010 Mar 15;50(6):805-13. doi: 10.1086/650573. PMID: 20166818; PMCID: PMC4696869.
Recommendations
The Use of Chest Ultrasound in Children with Parapneumonic Effusion Recommendation
In children with radiographic evidence of a moderate to large parapneumonic effusion, the panel suggests obtaining a chest ultrasound over CT or MRI to characterize the size and complexity of the effusion (conditional recommendation, very low certainty of evidence).
Remarks
- If chest ultrasound is unavailable, computed tomography (CT) or magnetic resonance imaging (MRI) of the chest may be performed to characterize the size and complexity of the effusion.
- In children with a small parapneumonic effusion on chest x-ray and/or with minimal respiratory symptoms, additional imaging (e.g., chest ultrasound, CT, or MRI) is generally not recommended.
The Use of Pleural Fluid Drainage versus Observation Recommendations
- In children with small, uncomplicated parapneumonic effusions, the IDSA panel suggests observation over pleural drainage (conditional recommendation, very low certainty of the evidence).
- In children with moderate parapneumonic effusions associated with respiratory distress, large parapneumonic effusions, or documented purulent effusions, the panel recommends pleural drainage (no new evidence) [2011 IDSA CAP guideline].
The Use of Pleural Fluid Drainage compared to Surgical Debridement Recommendation
In children and adolescents (3 months to 18 years) with pneumonia-associated empyema in whom pleural drainage is indicated, the panel suggests using chest tube drainage and intrapleural fibrinolytics rather than surgical debridement as first-line therapy in most cases (conditional recommendation, very low certainty of evidence).
Remarks
- Although similar outcomes are observed between chest tube placement (i.e., thoracostomy) with fibrinolytics and VATS, chest tube placement with fibrinolytics is less invasive, less costly, and can often be performed without general anesthesia.
- VATS may be reserved for a subset of patients, such as those with extensive loculation and those with refractory disease after chest tube placement with fibrinolysis.
- The decision on the approach to pleural drainage can be influenced by the availability of local resources and personnel.
The Choice of Chest Tube Size Recommendation
In children (3 months to 18 years) with parapneumonic effusion or empyema that necessitates drainage, the panel members suggest the use of small-bore (≤12Fr) chest tubes over large-bore (≥14FR) tubes (conditional recommendation, very low certainty of evidence).
Remarks
- Smaller tubes are effective at allowing for adequate drainage and for subsequent fibrinolysis.
- Since the last IDSA update, all published protocols used 12 Fr or smaller chest tubes.
The Use of tPa and DNase or tPa Alone for Fibrinolysis Recommendation
In children (3 months to 18 years of age) with pneumonia-associated empyema, the panel suggests administering tPA alone over tPA and DNase (conditional recommendation, low certainty of evidence).
A strong recommendation means most informed people would choose the recommended course of action and only a small proportion would not.
A conditional recommendation means the majority of informed people would choose the suggested course of action but many would not.
The Use of Chest Ultrasound in Children with Parapneumonic Effusion
Last Updated:
February 24, 2026Literature Review Conducted:
July 14, 2024This Recommendation Is Endorsed By The Pediatric Infectious Diseases Society (PIDS)
Authors:
Krow Ampofo*, Shawn D. St. Peter*, Thomas Brogan, Michael D. Cabana, Claudia Espinosa, Todd A. Florin, Jeffrey S. Gerber, Michelle Gill, Debra L. Palazzi, Mark Sawyer, Angela M. Statile, Derek J. Williams, Sheena Patel**, Samir S. Shah**, Mark I. Neuman**Keywords:
Community-acquired pneumonia, parapneumonic effusion, empyema, complicated pneumonia, chest ultrasound, computed tomographyRecommendation
[View full manuscript here]
Clinical Question
In children with parapneumonic effusion, should chest ultrasound or cross-sectional imaging (i.e., CT, MRI) be used to determine the character and confirm the size of the parapneumonic effusion?
Recommendation
In children with radiographic evidence of a moderate to large parapneumonic effusion, the panel suggests obtaining a chest ultrasound over CT or MRI to characterize the size and complexity of the effusion (conditional recommendation, very low certainty of evidence).
Remarks
- If chest ultrasound is unavailable, computed tomography (CT) or magnetic resonance imaging (MRI) of the chest may be performed to characterize the size and complexity of the effusion.
- In children with a small parapneumonic effusion on chest x-ray and/or with minimal respiratory symptoms, additional imaging (e.g., chest ultrasound, CT, or MRI) is generally not recommended.
A strong recommendation means most informed people would choose the recommended course of action and only a small proportion would not.
A conditional recommendation means the majority of informed people would choose the suggested course of action but many would not.
Results
A comprehensive literature search identified two observational studies assessing the accuracy of chest ultrasound in the evaluation of children with complicated pneumonia and parapneumonic effusion [Kurian, Thareeb]. In these studies, compared to chest CT (index test), the chest ultrasound sensitivity was 0.94 (95% CI: 0.87-1.00), and specificity was 1.00 (95% CI: 0.40 to 1.00).
Rationale for Recommendation(s)
For children needing further radiographic evaluation of parapneumonic effusion or empyema beyond a chest radiograph, a chest ultrasound can be used to guide further management. Compared with other imaging modalities, benefits of chest ultrasound include the lack of radiation exposure, and the ability to perform at the patient’s bedside, both for diagnostic monitoring (i.e., serial exams) and for assisting with therapeutic intervention (i.e., drainage). The panel members determined that contextual factors such as resources, feasibility, acceptability and equity for populations and health care systems across the United States favors the use of chest ultrasound.
References
- Kurian J, Levin TL, Han BK, Taragin BH, Weinstein S. Comparison of ultrasound and CT in the evaluation of pneumonia complicated by parapneumonic effusion in children. AJR Am J Roentgenol 2009; 193(6): 1648-54.
- Thareeb K, Zghair M, Hassan Q. Diagnostic Ability of Chest Ultrasound in Selective Paediatric Pneumonia Alternative to CT scan: A single-center Comparative Observational Study. Journal of Nepal Paediatric Society 2023; 42(03).
The Use of Pleural Fluid Drainage versus Observation
Last Updated:
February 24, 2026Literature Review Conducted:
August 22, 2024This Recommendation Is Endorsed By The Pediatric Infectious Diseases Society (PIDS)
Authors:
Shawn D. St. Peter*, Krow Ampofo*, Thomas Brogan, Michael D. Cabana, Claudia Espinosa, Todd A. Florin, Jeffrey S. Gerber, Michelle Gill, Debra L. Palazzi, Mark Sawyer, Angela M. Statile, Derek J. Williams, Sheena Patel**, Mark I. Neuman**, Samir S. Shah**Keywords:
Pneumonia, parapneumonic effusion, empyema, thoracentesisRecommendation
[View full manuscript here]
Clinical Question
In children (3 months to 18 years of age) with parapneumonic effusion, is pleural fluid drainage more beneficial than observation?
Recommendation 1
In children with small, uncomplicated parapneumonic effusions, the IDSA panel suggests observation over pleural drainage (conditional recommendation, very low certainty of the evidence).
Recommendation 2
In children with moderate parapneumonic effusions associated with respiratory distress, large parapneumonic effusions, or documented purulent effusions, the panel recommends pleural drainage (no new evidence) [2011 IDSA CAP guideline].
A strong recommendation means most informed people would choose the recommended course of action and only a small proportion would not.
A conditional recommendation means the majority of informed people would choose the suggested course of action but many would not.
Results
A comprehensive literature search identified three observational studies compared managements strategies of children with pleural effusions, including: 1) chest tube with or without intrapleural fibrinolysis, 2) video-assisted thoracoscopic surgery (VATS), or 3) antibiotics alone [Carter, Goldin, Segerer]. Two studies examined length of stay following pleural drainage procedures compared with observation alone [Carter, Segerer]. Combined data from these studies demonstrated that observation alone was associated with shorter length of hospital stay (mean difference -4.65 days; 95% CI -3.87 to -5.43) [6,8]. Goldin 2012 found no difference in mortality when comparing observation alone with pleural drainage (3.1% vs 3.3%, RR 0.93; 95% CI 0.78-1.11) [Goldin]. However, fluid drainage was associated with a reduced need for subsequent procedures compared to observation alone (26.2% vs. 45.0%; RR 1.71; 95% CI 1.38-2.13) [Segerer]. Goldin et al observed that waiting 1-3 days before drainage was associated with a lower mortality rate, while waiting >7 days was associated with a higher mortality rate [Goldin].
Rationale for Recommendation(s)
Given the results of the included studies, the panel conditionally recommends observation over drainage in children with small parapneumonic effusions. The panel considered costs, resources required and feasibility and determined that that these would probably favor observation.
Pleural drainage should be performed in children with large effusions. In these children, infection of the lung parenchyma, systemic response to infection, and a space occupying process around the lung may contribute to hemodynamic and respiratory instability.
References
- Carter E, Waldhausen J, Zhang W, Hoffman L, Redding G. Management of children with empyema: Pleural drainage is not always necessary. Pediatr Pulmonol 2010; 45(5): 475-80.
- Goldin AB, Parimi C, LaRiviere C, Garrison MM, Larison CL, Sawin RS. Outcomes associated with type of intervention and timing in complex pediatric empyema. Am J Surg 2012; 203(5): 665-73.
- Segerer FJ, Seeger K, Maier A, et al. Therapy of 645 children with parapneumonic effusion and empyema-A German nationwide surveillance study. Pediatr Pulmonol 2017; 52(4): 540-7.
The Use of Pleural Fluid Drainage compared to Surgical Debridement
Last Updated:
February 24, 2026Literature Review Conducted:
July 14, 2024This Recommendation Is Endorsed By The Pediatric Infectious Diseases Society (PIDS)
Authors:
Krow Ampofo*, Shawn D. St. Peter*, Thomas Brogan, Michael D. Cabana, Claudia Espinosa, Todd A. Florin, Jeffrey S. Gerber, Michelle Gill, Debra L. Palazzi, Mark Sawyer, Angela M. Statile, Derek J. Williams, Sheena Patel**, Samir S. Shah**, Mark I. Neuman**Keywords:
Empyema, pediatric, fibrinolysis, decorticationRecommendation
[View full manuscript here]
Clinical Question
In a child in whom pleural drainage is indicated, should chest tube placement with fibrinolysis be performed in preference to surgical debridement (i.e., Video-assisted thoracoscopic surgery)?
Recommendation
In children and adolescents (3 months to 18 years) with pneumonia-associated empyema in whom pleural drainage is indicated, the panel suggests using chest tube drainage and intrapleural fibrinolytics rather than surgical debridement as first-line therapy in most cases (conditional recommendation, very low certainty of evidence).
Remarks
- Although similar outcomes are observed between chest tube placement (i.e., thoracostomy) with fibrinolytics and VATS, chest tube placement with fibrinolytics is less invasive, less costly, and can often be performed without general anesthesia.
- VATS may be reserved for a subset of patients, such as those with extensive loculation and those with refractory disease after chest tube placement with fibrinolysis.
- The decision on the approach to pleural drainage can be influenced by the availability of local resources and personnel.
A strong recommendation means most informed people would choose the recommended course of action and only a small proportion would not.
A conditional recommendation means the majority of informed people would choose the suggested course of action but many would not.
Results
A comprehensive literature search identified seven (4 randomized, controlled and 3 observational) studies which compared chest tube drainage to VATS [Marhuenda, Sonnapa, St Peter, Cobanoglu, Griffith, Livingston, Shah]. Four randomized controlled trials compared chest tube drainage with fibrinolytics with thoracoscopy in children who presented with radiographic evidence of parapneumonic effusion or empyema with indication for drainage [Marhuenda, Sonnappa, St Peter, Cobanoglu]. Likewise, three retrospective cohort studies compared VATS debridement with chest tube drainage with fibrinolytics in children [Griffith, Livingston, Shah].
Surgical debridement was associated with 1.14 fewer days in the hospital than chest tube drainage with fibrinolytics; however, this difference was not found to be statistically significant (95% CI -3.38 to 1.10). Two observational studies also assessed length of stay, both of which found no significant differences between the surgical debridement and chest tube drainage [Griffith, Livingston].
The studies provide conflicting results regarding the need for further intervention. While 3 observational studies reported surgical debridement being associated with fewer subsequent procedures (10.3% vs 24.5%; RR 0.45; 95% CI 0.33-0.60) [Griffith, Livingston, Shah], 3 randomized controlled trials found no difference in the pooled risk of undergoing further interventions between surgical debridement and chest tube drainage (17.3% vs 16.8% RR 1.01; 95 CI 0.56 to 1.82) [Sonnappa, Cobanoglu].
Surgical debridement was associated with fewer chest tube days (MD -1.9; 95% CI -3.78 to 0.02) but no statistical difference in duration of supplemental oxygen requirement (MD -2.3; 95% CI -6.52 to 1.91).
Rationale for Recommendation(s)
While the studies demonstrated similar outcomes between surgical debridement and chest tube drainage with fibrinolytics, the panel conditionally suggests chest tube drainage with fibrinolysis over surgical debridement. The panel took into consideration the higher costs, feasibility and risks associated with surgical debridement in children. The panel discussed that some hospitals may not have the necessary expertise or resources to perform VATS, particularly in young children. Taken together, these factors favor the use of chest tube drainage with fibrinolysis.
References
- Marhuenda C, Barcelo C, Fuentes I, et al. Urokinase versus VATS for treatment of empyema: a randomized multicenter clinical trial. Pediatrics 2014; 134(5): e1301-7.
- Sonnappa S, Cohen G, Owens CM, et al. Comparison of urokinase and video-assisted thoracoscopic surgery for treatment of childhood empyema. Am J Respir Crit Care Med 2006; 174(2): 221-7.
- St Peter SD, Tsao K, Spilde TL, et al. Thoracoscopic decortication vs tube thoracostomy with fibrinolysis for empyema in children: a prospective, randomized trial. J Pediatr Surg 2009; 44(1): 106-11; discussion 11.
- Cobanoglu U, Sayir F, Bilici S, Melek M. Comparison of the methods of fibrinolysis by tube thoracostomy and thoracoscopic decortication in children with stage II and III empyema: a prospective randomized study. Pediatr Rep 2011; 3(4): e29.
- Griffith D, Boal M, Rogers T. Evolution of practice in the management of parapneumonic effusion and empyema in children. J Pediatr Surg 2018; 53(4): 644-6.
- Livingston MH, Colozza S, Vogt KN, Merritt N, Butter A. Making the transition from video-assisted thoracoscopic surgery to chest tube with fibrinolytics for empyema in children: Any change in outcomes? Can J Surg 2016; 59(3): 167-71.
- Shah SS, Hall M, Newland JG, et al. Comparative effectiveness of pleural drainage procedures for the treatment of complicated pneumonia in childhood. J Hosp Med 2011; 6(5): 256-63.
The Choice of Chest Tube Size
Last Updated:
February 24, 2026Literature Review Conducted:
July 14, 2024This Recommendation Is Endorsed By The Pediatric Infectious Diseases Society (PIDS)
Authors:
Shawn D. St. Peter*, Krow Ampofo*, Thomas Brogan, Michael D. Cabana, Claudia Espinosa, Todd A. Florin, Jeffrey S. Gerber, Michelle Gill, Debra L. Palazzi, Mark Sawyer, Angela M. Statile, Derek J. Williams, Sheena Patel**, Mark I. Neuman**, Samir S. Shah**Keywords:
Chest tube, thoracostomy, pediatric, parapneumonic effusionRecommendation
[View full manuscript here]
Clinical Question
In children with parapneumonic effusion, should small-bore (≤12Fr) or large-bore (≥14Fr) chest tubes be used for drainage?
Recommendation
In children (3 months to 18 years) with parapneumonic effusion or empyema that necessitates drainage, the panel members suggest the use of small-bore (≤12Fr) chest tubes over large-bore (≥14FR) tubes (conditional recommendation, very low certainty of evidence).
Remarks
- Smaller tubes are effective at allowing for adequate drainage and for subsequent fibrinolysis.
- Since the last IDSA update, all published protocols used 12 Fr or smaller chest tubes.
A strong recommendation means most informed people would choose the recommended course of action and only a small proportion would not.
A conditional recommendation means the majority of informed people would choose the suggested course of action but many would not.
Results
A comprehensive search identified a single observational study addressing the panel’s inclusion criteria [Lin 2011]. In this retrospective cohort study, 20 children were treated with traditional chest tubes (14-16Fr), and 12 were treated with smaller tubes (7-12 Fr). There was no statistically significant difference in length of stay between cohorts with different catheter sizes (12.5 ± 5.6 days in the traditional chest tube group vs. 17.3 ± 8.5 days in the smaller chest tube group, p=0.13). Additionally, no difference in the risk of adverse events or procedure-related complications were observed between groups.
Rationale for Recommendation(s)
Comparative evidence shows similar outcomes between small-bore and large-bore chest tubes, suggesting there are no disadvantages to using smaller tubes to achieve adequate drainage. Since there are no clear benefits to using larger-bore chest tubes, and because smaller tubes are less invasive and associated with less pain, small-bore chest tube is recommended.
References
- Lin CH, Lin Wc Fau - Chang J-S, Chang JS. Comparison of pigtail catheter with chest tube for drainage of parapneumonic effusion in children. 2011; (2212-1692 (Electronic)).
The Use of tPa and DNase or tPa Alone for Fibrinolysis
Last Updated:
February 24, 2026Literature Review Conducted:
July 14, 2024This Recommendation Is Endorsed By The Pediatric Infectious Diseases Society (PIDS)
Authors:
Krow Ampofo*, Shawn D. St. Peter*, Thomas Brogan, Michael D. Cabana, Claudia Espinosa, Todd A. Florin, Jeffrey S. Gerber, Michelle Gill, Debra L. Palazzi, Mark Sawyer, Angela M. Statile, Derek J. Williams, Sheena Patel**, Samir S. Shah**, Mark I. Neuman**Keywords:
Parapneumonic empyema, tissue plasminogen activator (tPA), dornase alfa (DNase)Recommendation
[View full manuscript here]
Clinical Question
In children (3 months to 18 years) with parapneumonic empyema, should tPA and DNase or tPA alone be used for fibrinolysis?
Recommendation
In children (3 months to 18 years of age) with pneumonia-associated empyema, the panel suggests administering tPA alone over tPA and DNase (conditional recommendation, low certainty of evidence).
A strong recommendation means most informed people would choose the recommended course of action and only a small proportion would not.
A conditional recommendation means the majority of informed people would choose the suggested course of action but many would not.
Results
A comprehensive literature search identified a single randomized controlled trial (RCT) that met the panel’s inclusion criteria [Livingston 2020]. In this study, a total of 97 children with parapneumonic empyema requiring pleural drainage were randomized to receive either tPA and placebo or tPA followed by DNase. Treatment with tPA and DNase was not associated with decreased hospital length of stay compared with tPA and placebo (mean [SD] length of stay, 9.0 [4.9] vs 9.1 [5.3] days; mean difference [MD] -0.1 days; 95%CI, −2.0 to 2.1; p=0.96). Similarly, no significant differences were observed for the need for additional intervention, such as repeat pleural drainage procedures (8% in the tPA plus DNase vs. 4% in the tPA plus placebo group; MD 4.0%; 95% CI -5.5 to 13.5; p=0.41). There were a similar number of adverse events across both groups (tPA and DNase, 24% vs tPA alone, 29%; p=0.64), and no deaths were reported in either group.
Rationale for Recommendation(s)
The panel conditionally suggests giving tPA alone over tPA and DNase. The panel discussed the cost of DNase as typically higher, and it is not approved for intrapleural use in the United States, which makes implementation less feasible. Further, the addition of DNase requires a second delivery of medication. If the pleural space is adequately evacuated with a chest tube, additional DNase is not necessary, increases costs and risks, including bleeding and pain. If fibrinous septations are the main barrier to drainage, liquefying pus with DNase without breaking down septations just redistributes thick fluid into walled-off loculation.
References
- Livingston MH, Mahant S, Connolly B, et al. Effectiveness of Intrapleural Tissue Plasminogen Activator and Dornase Alfa vs Tissue Plasminogen Activator Alone in Children with Pleural Empyema: A Randomized Clinical Trial. JAMA Pediatr 2020; 174(4): 332-40.
Notes
Acknowledgments
First, the panel would like to acknowledge the previous panel, under the leadership of John Bradley, for their work on the previous iteration of this larger guideline. The panel would like to acknowledge the contributions of Elena Guadagno, medical librarian, for the creation and execution of PICO-specific literature search(es). Rebecca Goldwater and Loretta Dzanya provided project coordination. The panel would also like to acknowledge the following organizations and selected external reviewers for their review of the draft manuscript: the American Academy of Pediatrics (AAP), the Pediatric Infectious Diseases Society (PIDS), Drs. Annabelle De St. Maurice, Jason Newland, Nanda Ramchandar, Pranavi Sreeramoju, and Surabhi (Sara) Vora.
Samir S. Shah is Chair of the pediatric CAP guideline panel. Mark Neuman is the Vice-Chair of the panel. Shawn St Peter and Krow Ampofo served as clinical leads for the questions addressed in this manuscript. Remaining panelists assisted with the conception and design of the analysis, interpretation of data, drafting and revising the recommendations and manuscript, and final approval of the recommendations and manuscript to be published. Nigar Sekercioglu and Sheena Patel, methodologists, were responsible for general project management, designing and performing the systematic review, and leading the panel according to the GRADE process.
Conflicts of Disclosure
Possible conflicts of interest. Evaluation of relationships as potential conflicts of interest is determined by a review process. The assessment of disclosed relationships for possible COIs is based on the relative weight of the financial relationship (i.e., monetary amount) and the relevance of the relationship (i.e., the degree to which an association might reasonably be interpreted by an independent observer as related to the topic or recommendation of consideration).
The following panelists have reported relationships with the indicated companies since 2018, when guideline development began; these relationships are unrelated to the topic of community-acquired pneumonia (CAP). K.K.A. served as a scientific advisor for Merck; served as an advisor for Janssen Pharmaceuticals; received research funding from the National Institute of Allergy and Infectious Diseases (NIAID), the Centers for Disease Control and Prevention (CDC), and the State Government of Utah; receives research funding from Merck. M.D.C. served as an advisor for BioGaia, Thermo Fisher, Genentech, Novartis, and Nestlé; engaged in activities with the International Scientific Association for Probiotics and Prebiotics (ISAPP); served as a member of the U.S. Preventive Services Task Force (USPSTF); served as chair of the American Academy of Pediatrics Committee on Pediatric Research; received research funding from the Agency for Healthcare Research and Quality (AHRQ). C.E. served as an advisor for Sanofi, Janssen Pharmaceuticals, and Gilead; received honoraria from Sanofi, Astellas, the Kentucky Rural Health Association and Medavera for collaboration on the writing and publication of a manuscript addressing the use of molecular techniques in respiratory infections; received research funding from AstraZeneca, Merck, Enanta Pharmaceuticals, Clinetic, the National Institutes of Health (NIH), Melinta Therapeutics, Novavax, and Moderna; receives research funding from AHRQ, the Florida Department of Health, and CDC; serves as a member of the American Academy of Pediatrics Committee of Infectious Diseases and as a Board Director (District III) for the AAP Florida Chapter. T.A.F. received honoraria from DiaSorin and Medscape/WebMD; served in editorial roles with Pediatric Research and Pediatric Emergency Care; served as a council member for the Society for Pediatric Research; received research funding from the National Heart, Lung, and Blood Institute (NHLBI) and the Patient-Centered Outcomes Research Institute (PCORI); served as strategy and operations officer for the Society for Pediatric Research; serves on the Pediatric Academic Societies Board of Directors. J.S.G. received research funding from AHRQ, NIH, CDC, and PCORI. M.G. served as president of the Society for Pediatric Research; served as a member of the Society for Pediatric Research Board of Directors; served as a member of the Pediatric Academic Societies Board of Directors; served as ambassador for North American Regional Societies for the Society for Pediatric Research; receives research funding from NIAID; receives research funding from the St. Louis Children’s Hospital Foundation. D.L.P. received royalties from UpToDate; received honoraria from the American Academy of Pediatrics: Pediatrics Review and Education Program (PREP) for Infectious Diseases, JAMA Pediatrics, and UpToDate; received honoraria from Lurie Children’s Hospital and the American Medical Association (AMA); received research funding from AHRQ and the Pew Charitable Trusts; owned intellectual property from Elsevier; served as associate editor for the Journal of the Pediatric Infectious Diseases Society (JPIDS) and the American Academy of Pediatrics PREP ID; serves on the Pediatric Infectious Diseases Society (PIDS) Board of Directors and Executive Committee; serves as president of PIDS. M.S. received research funding from the County of San Diego Health and Human Services Agency. A.M.S. received research funding from PCORI, the Children and Youth with Special Health Care Needs National Research Network (CYSHCNet), and the Gerber Foundation; reported a family relationship in which a spouse served as a consultant for echocardiography study interpretation for the Midwest Cardiovascular Research and Education (MCORE) Foundation. D.J.W. received research funding from AHRQ and NIH for work related to the Seattle-based Pediatric Health Information Partnership (P-HIP) initiative aimed at improving pediatric mental health care; serves on the Executive Council for the Pediatric Research in Inpatient Setting (PRIS) Network; serves on the Editorial Board for the Journal of Hospital Medicine; receives research funding from AHRQ and NIH. S.S.S. served in editorial roles with JAMA Pediatrics for the American Medical Association; served as senior deputy editor and then editor-in-chief for the Journal of Hospital Medicine for the Society of Hospital Medicine; served as associate editor for the Journal of the Pediatric Infectious Diseases Society; served as a Committee on Infectious Diseases member for the American Academy of Pediatrics; served as vice-chair for Pediatric Research in Inpatient Settings; received honoraria from the Society of Hospital Medicine; received research funding from NIAID, NHLBI, PCORI, and the Ohio Department of Public Health. M.N. served as associate editor for Pediatrics for the American Academy of Pediatrics; received research grants from Harvard University; serves as Co-Editor-in-Chief of Pediatric Emergency Care; receives honoraria from Wolters Kluwer for editorial roles with UpToDate and Pediatric Emergency Care; provides medical expert consultation, including medical record review and expert testimony in legal cases for law firms on issues not related to pneumonia.
The following panelists have reported relationships with the indicated companies since 2018, when guideline development began; these relationships are related to the topic of community-acquired pneumonia (CAP). K.K.A. served in advisory roles involving patient enrollment for clinical trials with Johnson & Johnson and Merck related to lumicitabine, tedizolid, ceftolozane/tazobactam, and bezlotoxumab (all such relationships are no longer active). D.L.P. served on a data safety monitoring board for Pfizer for a trial related to azithromycin; received honoraria from Medscape related to an educational podcast (relationship ended in 2024); owns intellectual property related to educational materials for Elsevier; receives other remuneration from the AMA. D.J.W. received research funding from NIH/NIAID for work related to Vanderbilt/ICE-CAP/R01AI125642 and Vanderbilt/VTEU/HHSN272201300023I, CDC for work related to Vanderbilt/NVSN/U01IP001063 and AHRQ for work related to R01HS029331, the Reducing Overuse of Antibiotics with Decision Support (ROADS) study aimed at testing the safety and effectiveness of clinical decision support to promote antibiotic stewardship in lower respiratory tract infections in children; received in-kind research support from bioMérieux for procalcitonin assays related to CAP decision-making (this relationship is no longer active); reported a family relationship in which a spouse is employed by Pfizer and the funds are paid directly to them. M.N. served as a member of the Guideline Development Group of the World Health Organization for the Diagnosis and Management of Pneumonia and Diarrhea in Children. All other authors reported no disclosures.
Publication Disclaimer
©IDSA<2026>. All rights reserved. No part of this publication may be reproduced, stored in a retrieval system, transmitted, used for text and data mining, or used for training artificial intelligence, in any form or by any means, without the prior permission in writing, or as expressly permitted by law, by license or under terms agreed with the appropriate reprographics rights organization. For commercial re-use, please contact reprints@oup.com for reprints and translation rights for reprints. All other permissions can be obtained through the Oxford University Press RightsLink service via the Permissions link for this paper in Clinical Infectious Diseases. For further information please contact journals.permissions@oup.com.
Update History
As of February 24, 2026, the Infectious Diseases Society of America, in collaboration with the Pediatric Infectious Diseases Society, has released updated recommendations for the management of community-acquired pneumonia in infants and children older than 3 months of age, replacing the previous guidelines last updated in 2011. The current guidelines are available on the IDSA website and will be published in Clinical Infectious Diseases at a later date.