Serum therapy was used extensively before the development of antibiotics and vaccines, pioneered over a century ago in the fight against diphtheria. The discovery that serum made from the blood of immunized animals, mostly horses, could neutralize toxins and inhibit the growth of some bacteria provided an intervention — even if imperfect — for several deadly diseases. However, in the 1940s, the success of antibiotics and the development of many vaccines compromised the enthusiasm for research in the use of passive infusions of serum to prevent or treat infectious diseases.
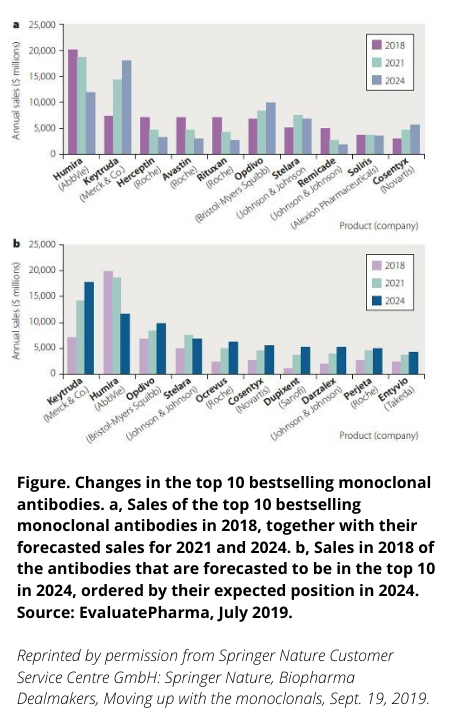
Georges Köhler and César Milstein won the Nobel Prize in 1984 for the development of monoclonal antibodies. Subsequently, the idea of using these agents for the treatment of cancers and a plethora of immunologic diseases took off (see figure).
Still, the infectious diseases community demonstrated limited interest in this approach (see the figure, where to date no antimicrobials have surfaced in the forecast, a situation that I believe SARS-CoV-2 will remedy rapidly), instead focusing on the development of ever broader antibacterial agents, and a growing number of important antiviral agents, especially those that would control the progression and transmission of HIV. To some extent the breadth and magnitude of infectious diseases seems inconsistent with mAb treatment, especially given the greater degree of difficulty in making, transporting and delivering these agents. Notable exceptions include the development of mAbs to prevent respiratory syncytial virus in premature newborns, mAbs to prevent and treat HIV, and mAbs to neutralize C. difficile infections.
A sea change in the use of antibodies
Witnessing the rapidity and severity of the COVID-19 pandemic — with no guarantee a vaccine could be developed — investigators immediately began to try convalescent plasma treatment, a logical exploration with considerable logistical challenges. At the same time, many established and “start-up” companies developed mAbs directed against different parts of the SARS-CoV-2 receptor binding domain. The ability to isolate clones of B cells responsible for antibodies discovered in patients recovering from SARS-CoV-2 (and the use of novel platforms informed by the viral structure) led to the rapid development of a plethora of mAbs that could neutralize the first circulating SARS-CoV-2 variants. These technologies arose in large part from decades of study of HIV, HIV vaccine development efforts and intensive study of HIV neutralizing mAbs.
In just a few months after the discovery of SARS-CoV-2, mAbs and mAb combinations entered prevention and treatment trials, ultimately leading to FDA emergency use authorizations. Neutralizing antibodies can provide an immediate and reliable benefit and are especially important for “compromised hosts” who might not have a satisfactory response to a SARS-CoV-2 vaccine or defenses to stop viral replication.
mAbs developed to treat cancer and immunologic diseases have many side effects, as they are directed at host tissues or host defenses. mAbs directed against viral pathogens have proven to be remarkably safe and well tolerated, and with extensive deployment in the COVID-19 pandemic, serious side effects have rarely been observed.
Unfortunately, SARS-CoV-2 has proven a very facile foe, and the evolution of mutant variants rapidly compromised most of the first crop of mAbs. These variants arise through “in host” selective pressures on the virus when it is still replicating and can be transmitted; de facto the virus must be exposed to autologous antibodies, with the same or similar sites targeted by mAbs. Currently, the only mAbs available for the SARS-CoV-2 Omicron variants now prevalent are the combination of tixagevimab and cilgavimab for prevention of infection, and bebtolivimab for treatment to prevent progression of COVID-19 in high-risk patients. All earlier mAbs approved for the SARS-CoV-2 variants that preceded the Omicron variant have lost required activity (at least in vitro) and EUA approval.
Tools for infectious disease prevention and treatment
Although the rise and fall of the first generation of mAbs directed against SARS CoV-2 has been challenging, the work has served as a critical proof of concept and catalyst. mAbs are tools that can play an important and unfilled role in infectious disease prevention and treatment. For SARS-CoV-2, targets will be identified that are unlikely to escape combinations of antibodies, regardless of mutations that occur.
In the future, we can expect the COVID-19 experience to galvanize development of mAbs for many other infectious diseases for which we have no vaccine, prophylaxis or treatment. We are limited only by our creativity. Looking ahead, the cost and logistical difficulties will decrease as mAbs are more widely used in infectious disease, as has been the case in the fields of oncology and rheumatology. mAbs will surface as an alternative strategy to traditional “small molecule” anti-infective agents, and passive immunity will serve as a true alternative to some vaccines. Stay tuned.